
Dr. Orgill is Vice Chairman for Quality Improvement in the Department of Surgery at Brigham and Women’s Hospital and Professor of Surgery at Harvard Medical School. He is a reconstructive plastic surgeon and has a PhD from MIT in Medical Engineering. He is the Director of the Brigham and Women’s Hospital Wound Care Center and runs a tissue engineering and wound healing Laboratory. His lab at BWH is working to develop better technologies to treat wounds including work with artificial skin, micromechanical forces, platelets and stem cells. He has consulted for several medical device and start-up companies and is the inventor on several patents. He worked on the team that developed Integra, a skin replacement therapy that has been commercially developed and used successfully on thousands of patients.
Orgill_Current Dialogues in Wound Management_2020_Article 1
As the number of wounds around the world continues to increase, devising paradigms to organize our clinical approach to accurately diagnose why wounds do not heal can be very helpful. A critical factor that contributes to wound healing is the perfusion of blood in the tissues around the wound.
Basic physiology
The body is carefully designed so that metabolically active cells are only within a fraction of a millimeter of a capillary.1 Capillaries are the small tubes that transport single blood cells (which carry critical oxygen), proteins, glucose and electrolytes in this amazing network. These networks also take away metabolic waste products including carbon dioxide and urea.2
In a typical adult, the heart pumps 5 liters of blood per minute through the circulatory system which includes 60,000 miles of capillary networks.3 Blood is driven across this vast network by the pressure generated by the heart. Blood pressure typically drops across a capillary network from about 35 mmHg to 18 mmHg.4 The skin has a large capillary density that is present in the superficial and deep dermal plexus of the dermis.5 The epidermis has no capillary networks but is instead nourished through diffusion from the dermis. As cells get further from the blood source, they tend to undergo programmed cell death, become desiccated and form the stratum corneum – the layer of skin that protects our internal organs from bacterial invasion and water loss.4
How does Perfusion Influence Wound Healing?
The most obvious example of a lack of perfusion is in individuals with a lack of arterial perfusion (Figure 1). Quite simply, there are blockages in the large arteries proximal to the wound that impede blood flow and reduce the perfusion pressure differential across capillary beds.6 As a result, there is a decrease in blood flow to these areas. When blood flow becomes too low, cells in the area begin to suffer from a lack of oxygen (hypoxia) which can lead to ischemia.7 If the hypoxia is not reversed, it can lead to cell death.8
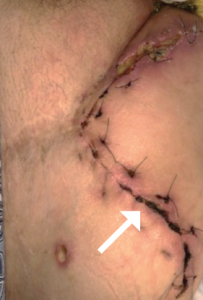
Loss of Perfusion Due to Venous Insufficiency
Similarly, there can be a loss of a perfusion differential across a capillary bed due to venous stasis, where the final venous pressure can be elevated.9 When this happens, blood can extravasate out the vessels and stain the skin (hemosiderin deposits) (Figure 2).
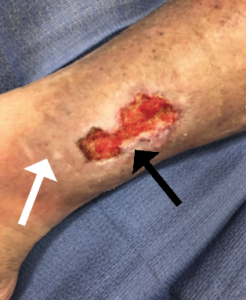
Lack of Mass Transport Due to Reduced Diffusiony
Many molecules are transported throughout the body through the capillary network until they reach their destination via the process known as diffusion.10 Basic diffusion equations have demonstrated that the amount of transport is related to the differences in concentration but is also related to the inverse square relationship between the distance the molecule must transfer.11 So, in states such as lymphedema when the extracellular space is filled with fluid, diffusion is slowed (Figure 3). Similarly, in radiation therapy, where the capillary density in tissues can be diminished, the diffusion of nutrients can be slowed.
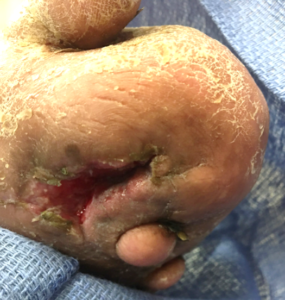
The Effects of Pressure on Perfusion
In diabetic foot ulcerations and in pressure injuries, prolonged pressure on the skin leads to a lack of perfusion in the soft tissues.12 If this occurs for longer than 3 to 4 hours, irreversible ischemia can occur leading to necrosis of the tissue.13 Very often, patients with diabetes have neuropathy making it difficult to feel pain if they injure their foot. Similarly, paraplegics or those with neurological deficits cannot feel or react to prolonged pressure to bony prominences such as the ischium or sacrum. After time, tissue necrosis can occur that can lead to ulceration.
Measurement of Perfusion
There are many methods to directly or indirectly measure perfusion. When blood is well oxygenated, it turns bright red and when it loses the oxygen molecules, it becomes dark red. Pulse oximetry can reliably measure these differences with fast response times and can show a beat-to-beat variability in oxygenation. Transcutaneous oxygen levels sample the oxygen that diffuses across the skin and can be useful in monitoring patients on high inspired oxygen. For patients with vascular disease, non-invasive vascular tests that include blood pressure measurements at various levels of the extremity as well as pulse volume recordings can give a quantifiable index of perfusion. For patients where more anatomic information is needed, injecting dye into the vascular system and imaging this with an x-ray, computed tomography scan and/or magnetic resonance imaging can be quite useful. Ultrasound is also a very effective method of assessing the performance of the heart.
For the skin, there are a variety of technologies that have been developed to measure perfusion.14 The simplest is to just look at the skin and use clinical judgement to know when skin perfusion changes. However, most clinicians prefer quantifiable and reproducible methods that are not clinician dependent. One common method used today is indocyanine green perfusion where fluorescent images of the skin can be obtained to gauge the arterial perfusion of skin flaps. We have found this to be effective in judging the long-term viability of mastectomy flaps in breast reconstruction.15 Another method that is becoming more popular is hyperspectral imaging, which allows for several wavelengths of light to shine on the skin and the spectra of the returning light is analyzed to calculate oxyhemoglobin and deoxyhemoglobin in the skin (Figure 4).16 These devices can be used without any need to inject dye.
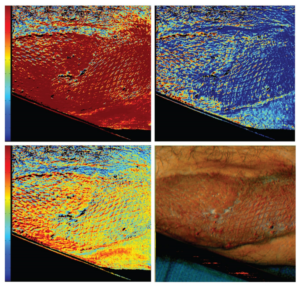
Conclusions
Perfusion is critical in allowing healthy tissue to survive and is also critical in contributing to wound healing. Arterial and venous stasis wounds have healing deficits because of perfusion. Radiation wounds and wounds from lymphedema have perfusion deficits because of lack of diffusion. Pressure injuries and diabetic foot ulcers can result from a lack of perfusion to the affected tissues. New methods to monitor perfusion give us renewed hope that better diagnosis, prevention and treatment of chronic wounds is a distinct possibility that we, as clinicians, can use in our armamentarium.
Patient photos courtesy of Dr. Dennis Orgill, Brigham and Women’s Hospital, Boston, MA.
References
- Pittman RN. Regulation of tissue oxygenation. In: Granger DN, Granger JP, eds. Colloquium Series on Integrated Systems Physiology: from Molecule to Function to Disease. San Rafael, CA: Morgan & Claypool Life Sciences; 2011:Vol 3 No 3:1-100.
- Institute for Quality and Efficiency in Health Care. What does blood do? InformedHealth.org website. https://www.ncbi.nlm.nih.gov/books/NBK279392/. Accessed March 7, 2020.
- Zimmermann KA. The Circulatory System: An amazing circuit that keeps our bodies going. LiveScience. https://www.livescience.com/22486-circulatory-system.html. Accessed March 7, 2020.
- OpenStax. Anatomy & Physiology. OpenStax CNX. http://cnx.org/contents/14fb4ad7-39a1-4eee-ab6e-3ef2482e3e22@8.24. Accessed March 7, 2020.
- Braverman IM. The cutaneous microcirculation. J Invest Derm Symp Proc. 2000;5(1):3-9.
- De Graaff JC, Ubbink DT, van der Spruit JA, Lagarde SM, Jacobs MJ. Influence of peripheral arterial disease on capillary pressure in the foot. J Vasc Surg. 2003;38(5): 1067-1074.
- Simon F, Oberhuber A, Floros N, et al. Acute limb ischemia – Much more than just a lack of oxygen. Int J Mol Sci. 2018;19(2):e374.
- Warren DR, Partridge M. The role of necrosis, acute hypoxia and chronic hypoxia in F-FMISO PET image contrast: a computational modelling study. Phys Med Biol. 2016;61(24): 8596-8624.
- Caprini JA, Partsch H, Simman R. Venous ulcers. J Am Coll Clin Wound Spec. 2012;4(3): 54-60.
- Popel AS. Analysis of capillary-tissue diffusion in multicapillary systems. Math Biosci. 1978;39(3-4):187-211.
- Crank J. The mathematics of diffusion. 2nd ed. Oxford, England. Oxford University Press. 1975.
- Liao F, Burns S, Jan YK. Skin blood flow dynamics and its role in pressure ulcers. J Tissue Viability. 2013;22(2):25-36.
- Kalogeris T, Baines CP, Krenz M, Korthuis RJ. Ischemia/Reperfusion. Compr Physiol. 2016;7(1):113-170.
- Bader DL, Worsley PR. Technologies to monitor the health of loaded skin tissues. BioMedical Eng Online. 2018;17(1):40.
- Griffiths M, Chae MP, Rozen WM. Indocyanine green-based fluorescent angiography in breast reconstruction. Gland Surg. 2016;5(2):133-149.
- Chin MS, Freniere BB, Lancerotto L, et al. Hyperspectral imaging as an early biomarker for radiation exposure and microcirculatory damage. Front Oncol. 2015;5:232.
@Copyright 2020 3M. All rights reserved. 3M and the other marks shown are marks and/or registered marks. Unauthorized use prohibited. PRA-PM-US-02322 (03/20).

